مواصفات المنتج
| الماركة | DAAN GENE CO., LTD. OF SUN YAT-SEN UNIVERSITY |
| الحالة | New |
| الكفالة | YES |
مواصفات المنتج
【INTENDED USE】
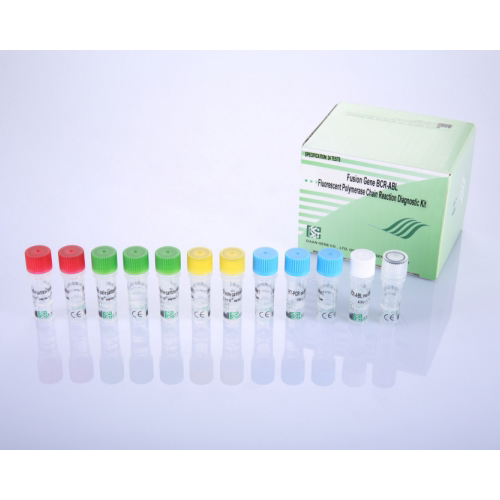
This kit is applicable to quantitative detection of Fusion Gene BCR-ABL expression in hematological system tumors such as chronic myelocytic leukemia (CML) and acute lymphocytic leukemia (ALL), etc., which has great guidance significance for clinical identification diagnosis, monitoring minimal residual disease relapse and evaluation of chemotherapy treatment effect, etc. The result is for clinical reference only, should not be a basis of making a final diagnosis or for discarding cases.
【PRINCIPLE】
According to Fusion Gene BCR-ABL and control gene TBP, this kit respectively designs specific primer and specific fluorescence probe of both genes, fitted with components of BCR-ABL reaction solution, TBP reaction solution, RT-PCR enzyme mixture, etc. Directly uses RNA as template for in vitro amplification with PCR-Fluorescence technology to detect Fusion Gene BCR-ABL mRNA expression levels.
【PERFORMANCE CHARACTERISTICS】
Sensitivity: 50 copies per µl
Linear Range: 1.0 x 103 to 1.0 x 108 copies per µl
Specification: 24 tests per kit
Shelf life: 9 months
Storage and Transportation: Stable at 2-8°C during transportation, must be stored at -20±5°C upon receipt.



 طريقة دفعة آمنة وموثوقة باستخدام منصة عبرة للدفع عبر الانترنت
طريقة دفعة آمنة وموثوقة باستخدام منصة عبرة للدفع عبر الانترنت  \
\